This is the Lewis structure for diethyl ether with a VSPER form of bent. It is C4H10O. Each oxygen to carbon bond is moderately covalent while both a carbon to carbon and a carbon to hydrogen are extremely covalent.
|
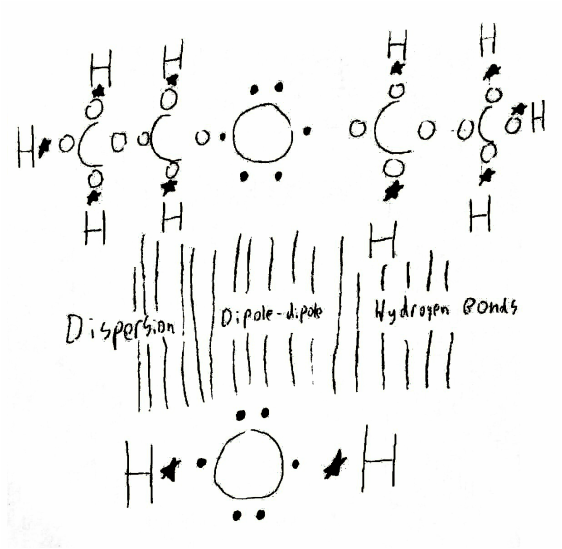
This is the attractions between one molecule of diethyl ether and one molecule of water. Three forces are in effect, dispersion, dipole-dipole, and hydrogen bonds. Dispersion is in effect because it occurs between any two adjacent molecules. Since both diethyl ether and water are polar, dipole-dipole is in effect since it occurs between any two polar molecules. Hydrogen bonds occur because the hydrogens in both molecules are attracted to the oxygen in the other. This is because hydrogen bonding is the attraction between hydrogen and oxygen, nitrogen, or fluorine.
|